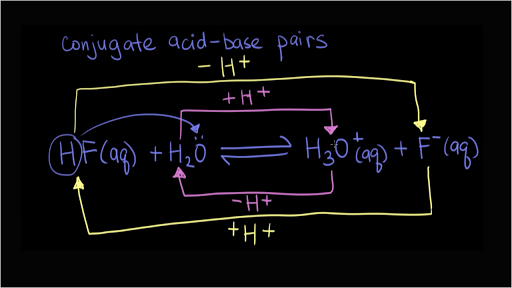
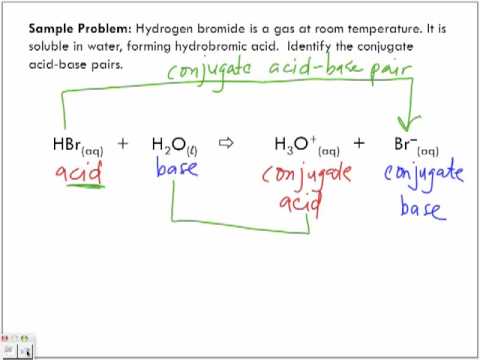
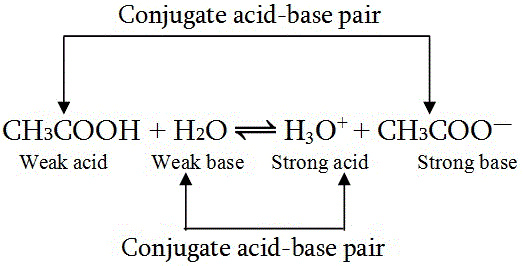
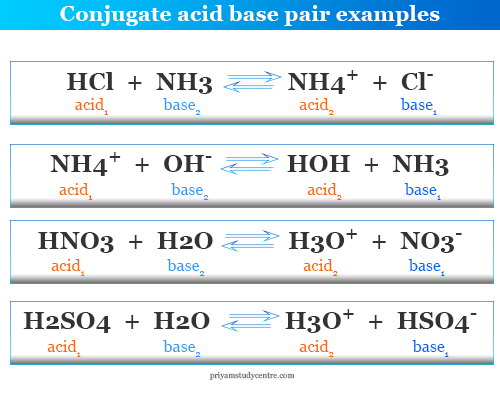
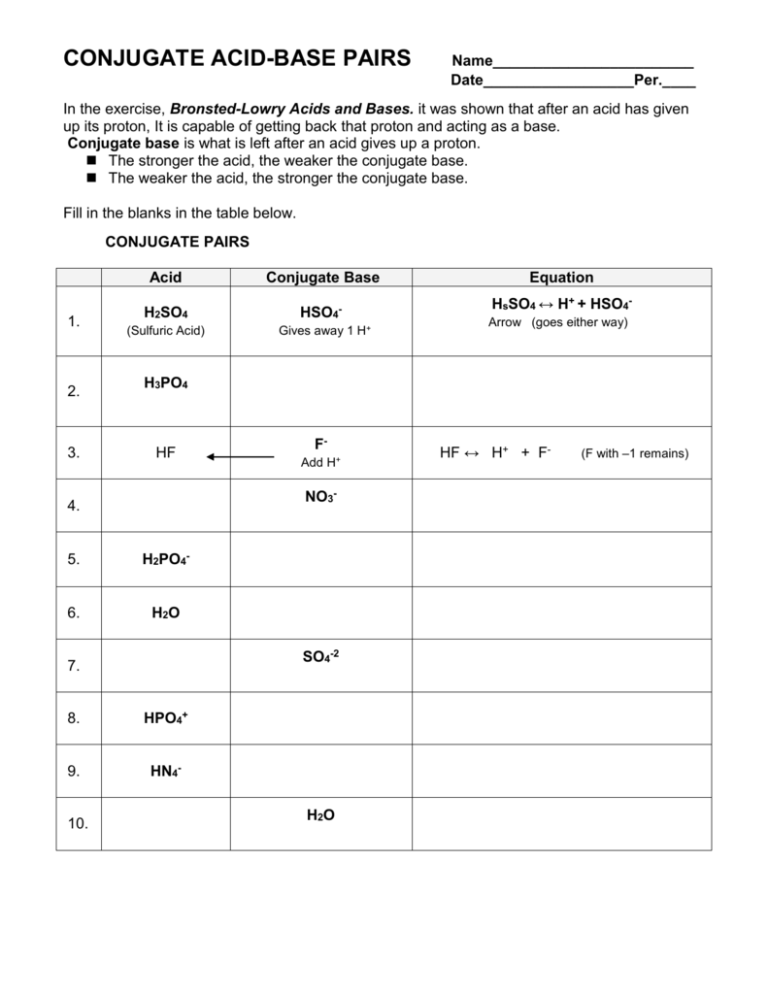
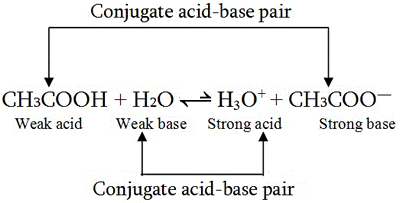
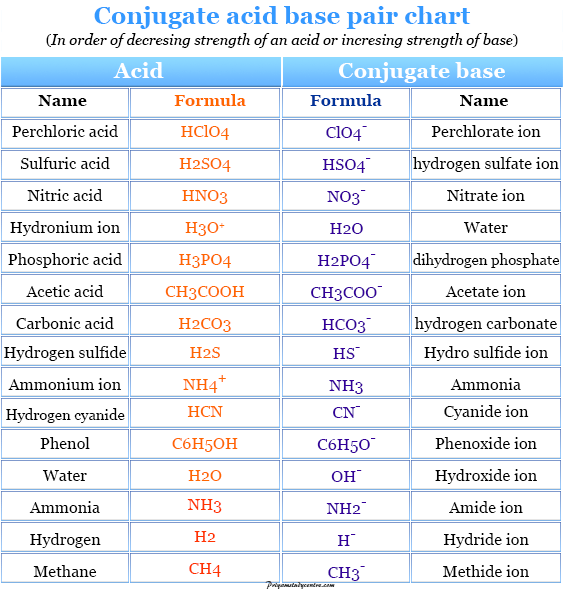
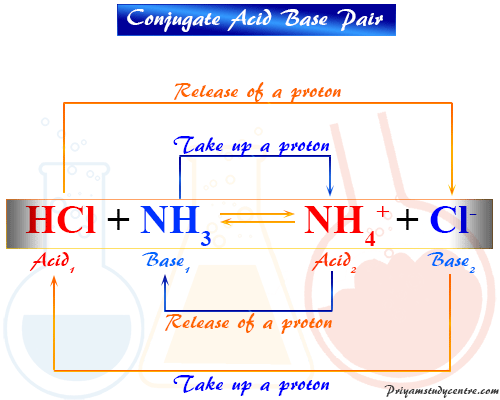
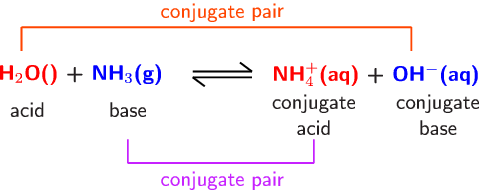
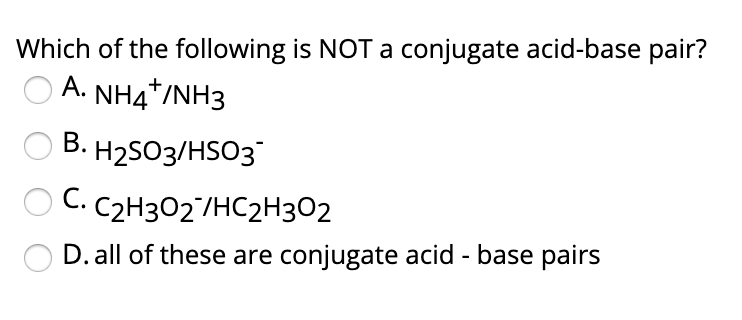Identify the conjugate acid-base pairs for the reaction (with the acid written first). CN- + H2O = HCN + OH- |CN- / HCN |HCN / CN- |OH- / H2O |H2O / OH- | Homework.Study.com

What is meant by the conjugate acid-base pair? Find the conjugate acid/ base for the following species: HNO 2 , CN - , HClO 4 , F - , OH - , CO 3 2- and S 2-