SOLVED: Predict the minor and major product DIPEA-diisopropylethylamina or Hunig s base DcJe malc prcducl rnE prcducl Predict products and indicate which one major product 4meducl ShowIne machansm oline lollowing reaclions Clearly
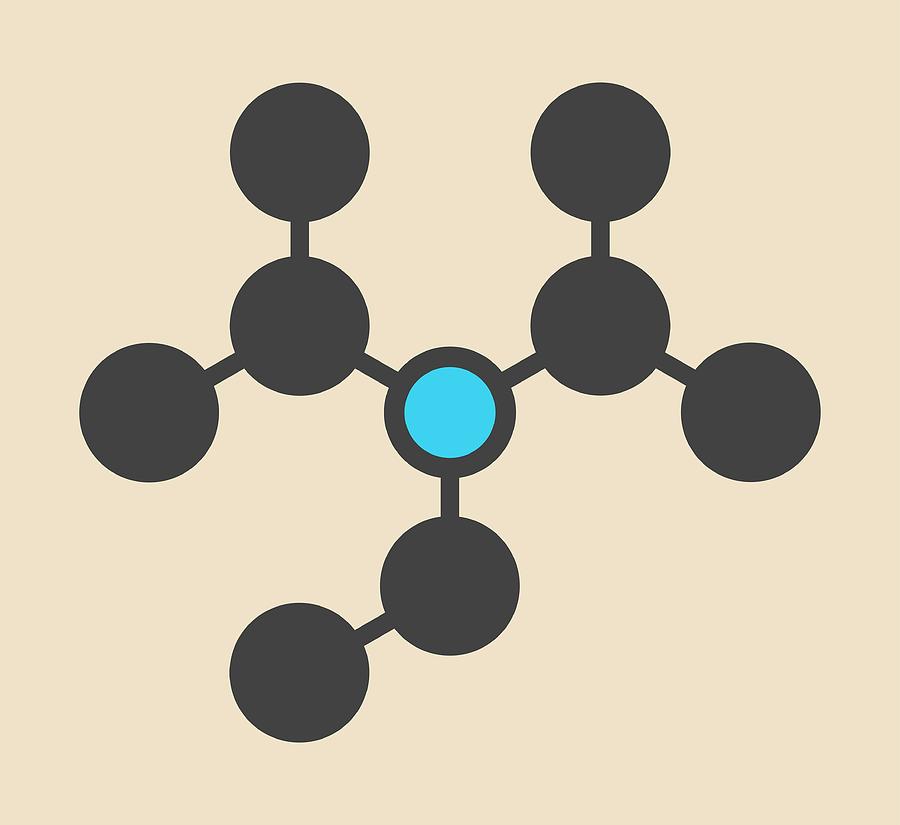
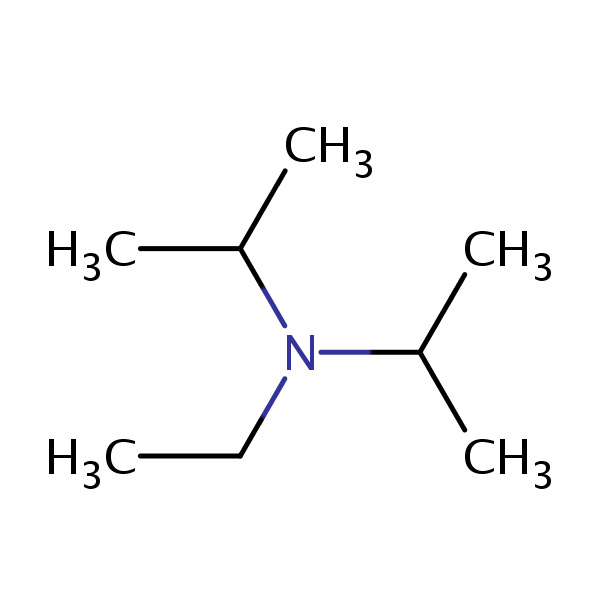
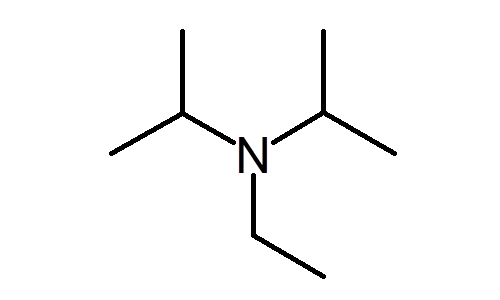
DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey Stock Photo - Alamy
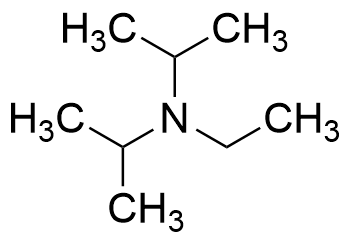
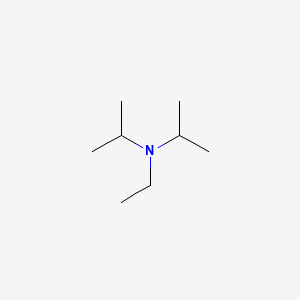
7087-68-5 | N,N-Diisopropylethylamine | 1,1'-Dimethyltriethylamine; Bis(1-methylethyl)ethylamine; DIEA; DIPEA; Diisopropylethylamine; Ethyl-N,N-diisopropylamine; Ethyldiisopropylamine; Huenig's base; Hunig's base; Hunig's reagent; N,N-Bis(1-methylethyl ...

DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey Stock Photo - Alamy

DIPEA-Promoted Reaction of 2-Nitrochalcones with Elemental Sulfur: An Unusual Approach to 2-Benzoylbenzothiophenes | Organic Letters
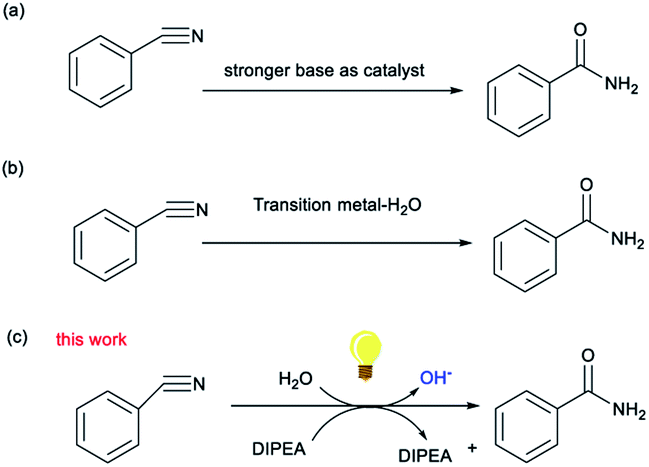
DIPEA-induced activation of OH − for the synthesis of amides via photocatalysis - RSC Advances (RSC Publishing) DOI:10.1039/D2RA02107B

Electrochemical Formation of Cinnamaldehyde by the Electrolyte System N,N‐ Diisopropylethylamine and 1,1,1,3,3,3‐Hexafluoropropan‐2‐ol - Imada - 2020 - ChemElectroChem - Wiley Online Library