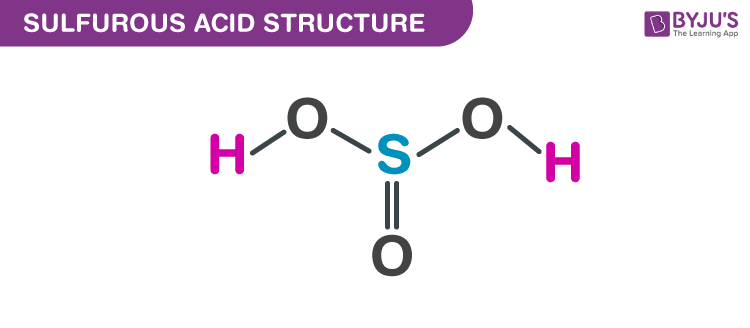
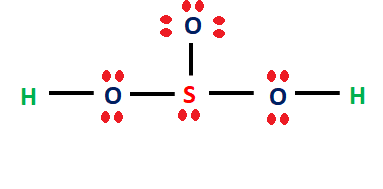
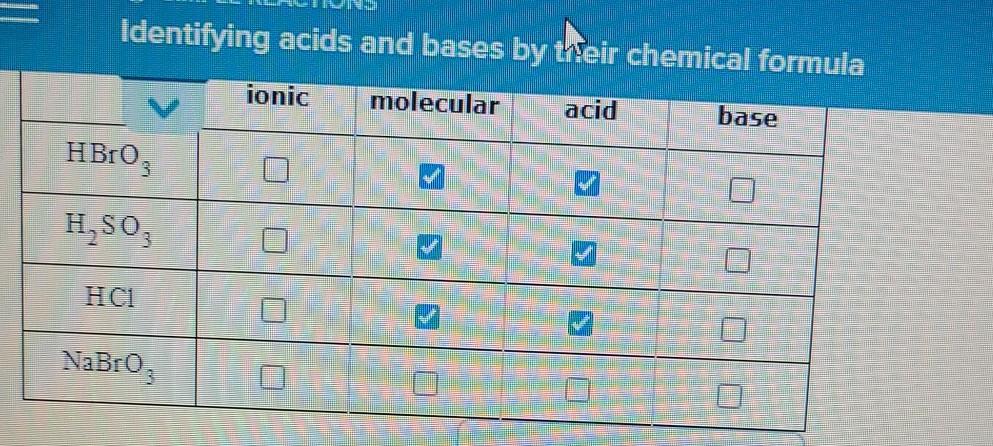
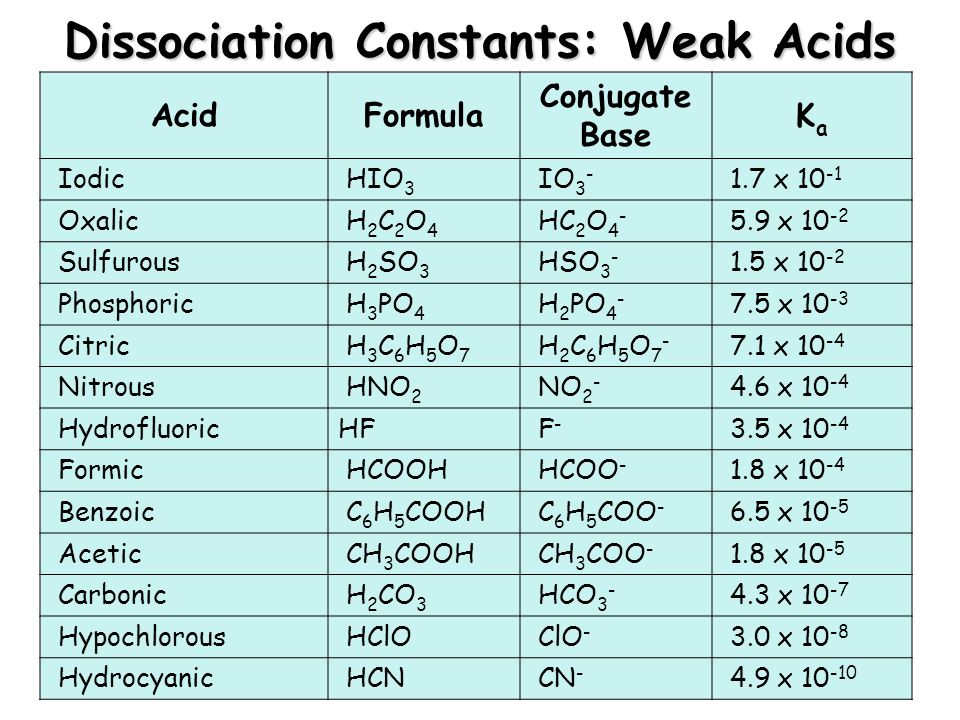
Acid-Base Test Review Definitions of Acids and Bases 1. Which of the following are Arrhenius acids? a. H2O b. H3PO4 c. NH3 d. H2SO3 2. Which of the. - ppt download
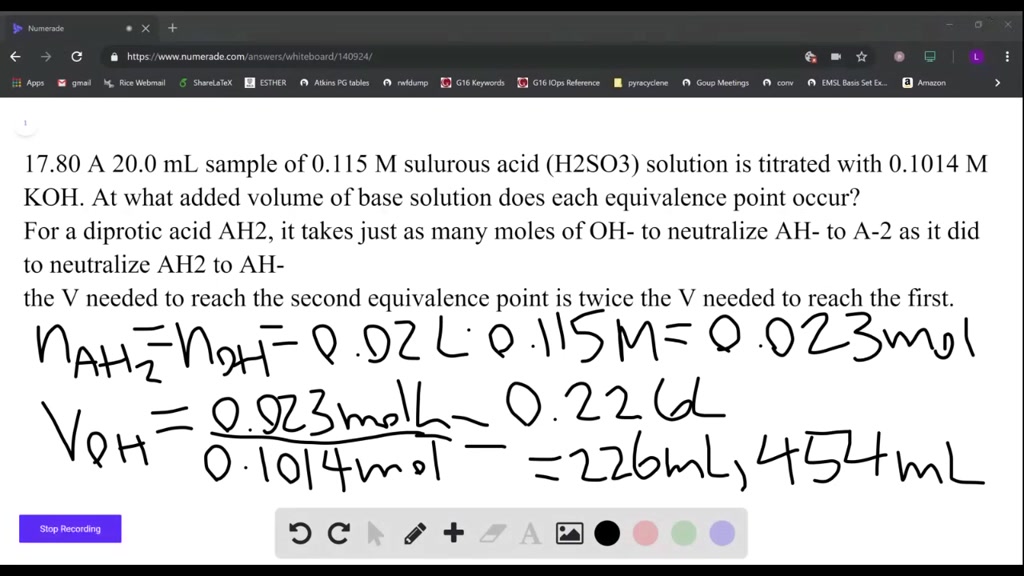
SOLVED:A 20.0 -mL sample of 0.115 M sulfurous acid (H2 SO3) solution is titrated with 0.1014 M KOH . At what added volume of base solution does each equivalence point occur?

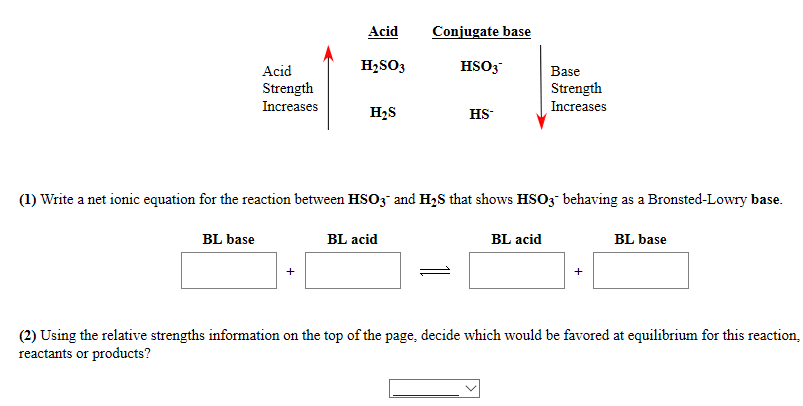
SOLVED: Konsider the reaction: H2SO3 HCO3" H2CO3 HSO3" a) Identify the acid, base, conjugate acid and conjugate base b. What is the conjugate base of water? C What is the value of [