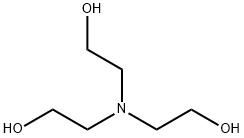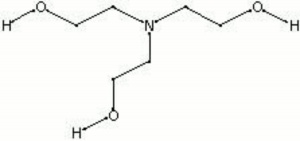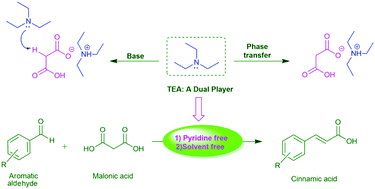
Triethylamine: a potential N-base surrogate for pyridine in Knoevenagel condensation of aromatic aldehydes and malonic acid - New Journal of Chemistry (RSC Publishing)

Synthesis and properties of triethanolamine-based salts with mineral and organic acids as protic ionic liquids - ScienceDirect

Scheme 1. Schematic view of the structures of Schiff-base ligands and... | Download Scientific Diagram

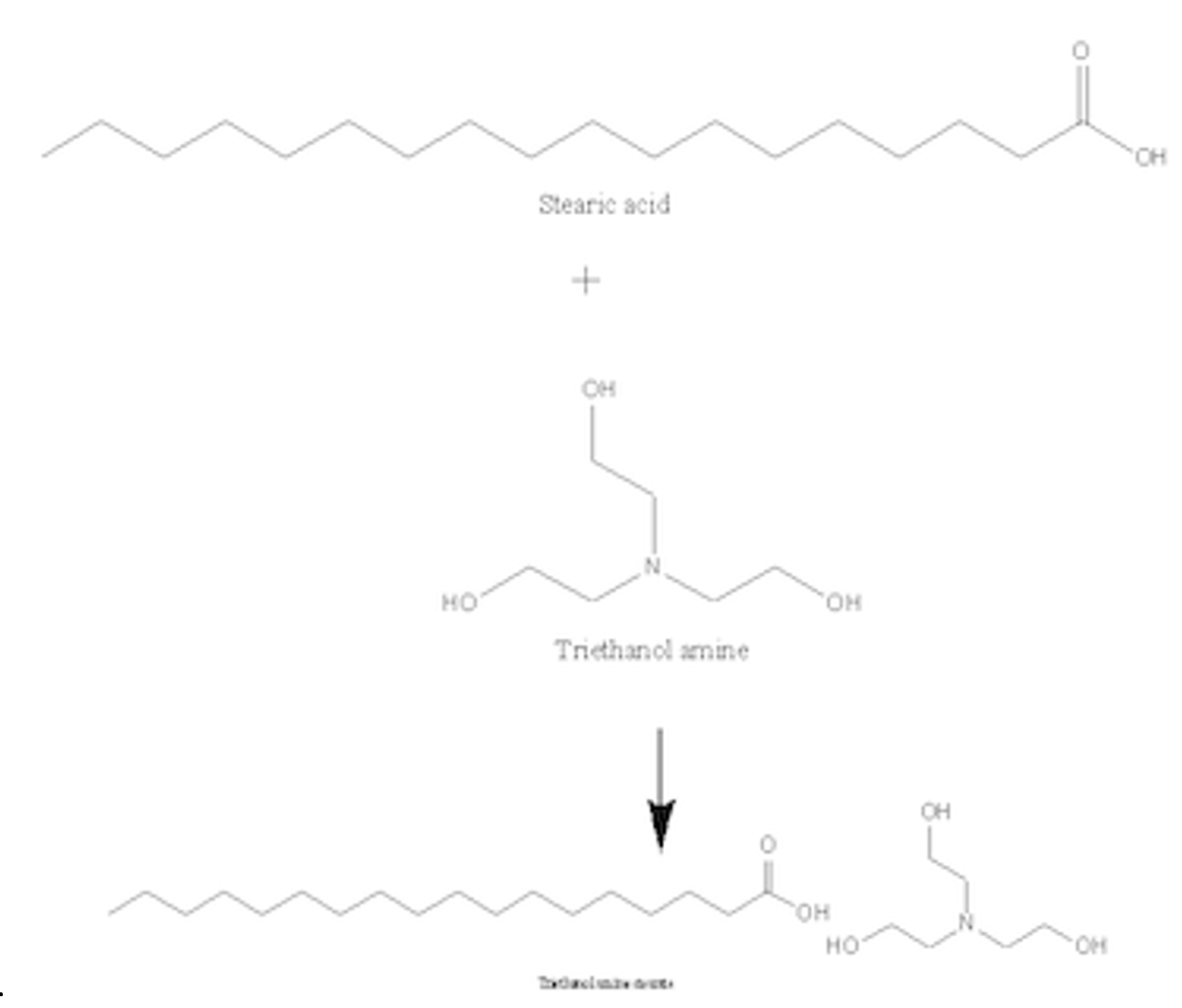
Triethanolamine as an Efficient and Reusable Base, Ligand and Reaction Medium for Phosphane‐Free Palladium‐Catalyzed Heck Reactions - Li - 2006 - European Journal of Organic Chemistry - Wiley Online Library

Triethanolamine as an Efficient and Reusable Base, Ligand and Reaction Medium for Phosphane‐Free Palladium‐Catalyzed Heck Reactions - Li - 2006 - European Journal of Organic Chemistry - Wiley Online Library
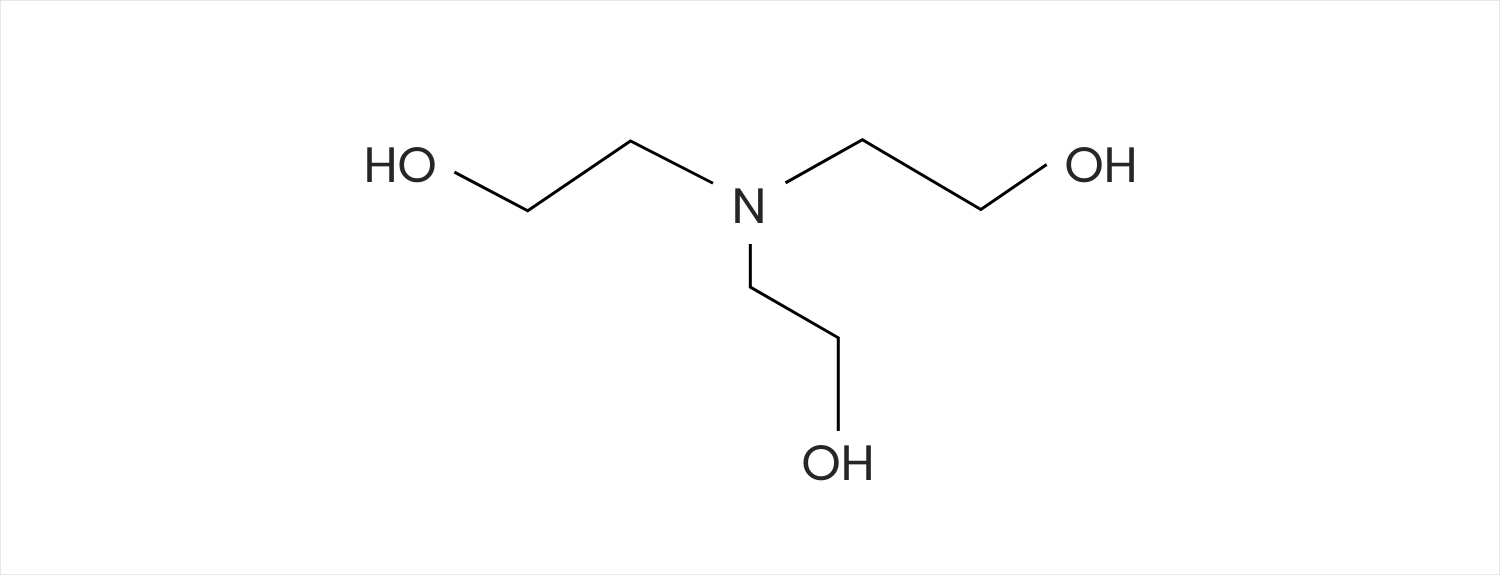
Triethanolamine - The Importance Of Non-Active Ingredients In Your Skincare - The Dermatology Review

Triethanolamine-based protic ionic liquids with various sulfonic acids: Synthesis and properties - ScienceDirect

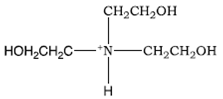
![T23040-1.0 - Triethanolamine, Free Base [2,2',2"-Nitrilothriethanol], 1 Liter T23040-1.0 - Triethanolamine, Free Base [2,2',2"-Nitrilothriethanol], 1 Liter](https://d2gdaxkudte5p.cloudfront.net/system/images/plabel_14881_20230721-085650.jpg)
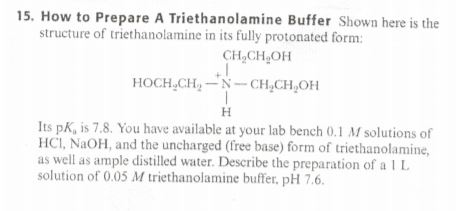





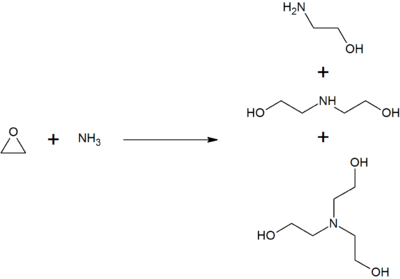
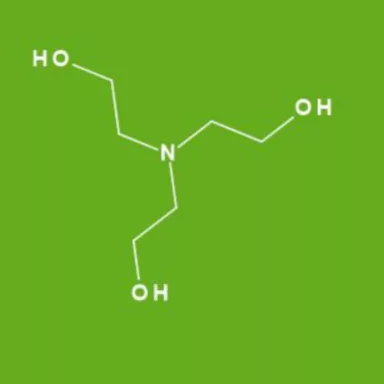

![T23040-4.0 - Triethanolamine, Free Base [2,2',2"-Nitrilothriethanol], 4 Liter T23040-4.0 - Triethanolamine, Free Base [2,2',2"-Nitrilothriethanol], 4 Liter](https://d2gdaxkudte5p.cloudfront.net/system/images/T23040-4.0_.jpg)
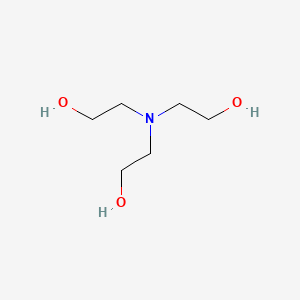

![T23040-1.0 - Triethanolamine, Free Base [2,2',2"-Nitrilothriethanol], 1 Liter T23040-1.0 - Triethanolamine, Free Base [2,2',2"-Nitrilothriethanol], 1 Liter](https://d2gdaxkudte5p.cloudfront.net/system/images/T23040-1.0_4.jpg)